|
1/15/2024 0 Comments Lithium battery anode cathode
The researchers note they could produce the new coating on these copper current collectors using standard fabrication techniques. The new battery can hold 72 percent more energy by weight and 95 percent more energy by volume than commercial lithium-ion batteries. “It’s like having an engine that is covered by a thick layer of rust on the inside,” he says. They also include lumps of “dead metal” and crusts of honeycomb-like material that impeded the battery’s performance. These include spiky dendrites that “can, and will, lead to battery short circuits between the anode and cathode, which in turn can cause battery fires,” Mitlin says. Without this new coating, the researchers found the copper foil became covered with irregular microscopic structures during recharging and discharging. They found this new coating helped lithium metal deposit and dissolve from the copper current collector in a thin uniform layer. The aim was to control the way in which lithium metal spread across or “wetted” the copper. 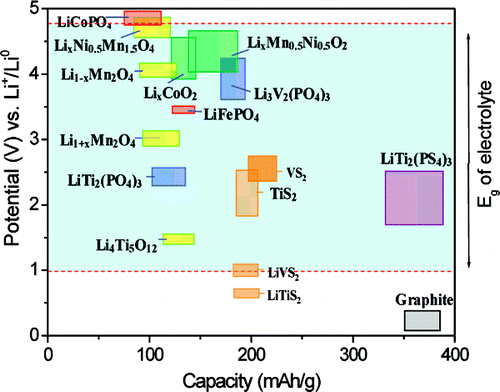
They explored coating its copper current collector with ultrathin lithium-activated tellurium. The scientists experimented with an anode-free, all-solid-state battery with a sulfide-based solid electrolyte. Now, in a new study, researchers show a new coating may overcome this problem. However, a key challenge that research on anode-free, all-solid-state batteries has faced is the problem they face going through cycles of discharging and recharging in a stable way. Using less material also reduces their cost, he adds. ”You are in effect eliminating half of the battery’s internal material,” says study senior author David Mitlin, a materials scientist at the University of Texas at Austin. This strategy could significantly boost how much energy these batteries might hold. One way scientists have sought to improve all-solid-state batteries is by replacing their conventional graphite anodes solely with a copper-foil current collector. However, the sulfides in these electrolytes can react with both electrodes, generating compounds that impede the flow of electricity within the batteries. For example, previous research found sulfide-based solid electrolytes could help create batteries that can store a lot of energy. Much remains uncertain when it comes to the best way to create a stable, useful, all-solid-state battery. In addition, all-solid-state lithium batteries are much safer than their conventional counterparts, which use organic liquid electrolytes that are typically flammable. This means that all-solid-state batteries can produce more energy than conventional batteries for the same amount of weight or space. Solid electrolytes are more compact than liquid or gel electrolytes. All-solid-state batteries instead employ solid electrolytes made of materials such as ceramics. The electrodes in lithium-ion batteries typically interact through liquid or gel electrolytes. These electrode materials are coated onto metal foils that collect the generated current-for the anode, this metal is often copper, and for the cathode, aluminum. The anode is often comprised of graphite, while the cathode is very often a metal oxide. A typical lithium-ion battery’s electrodes are made of substances whose structures can store and release electrically charged lithium ions. The secret behind this novel prototype’s success? Getting rid of one of a battery’s usual electrodes, a new study finds.Ĭonventional batteries supply electricity via chemical reactions between two electrodes-the negatively charged anode, where electrons flow out of a battery, and the positively charged cathode, where electrons can enter a battery. 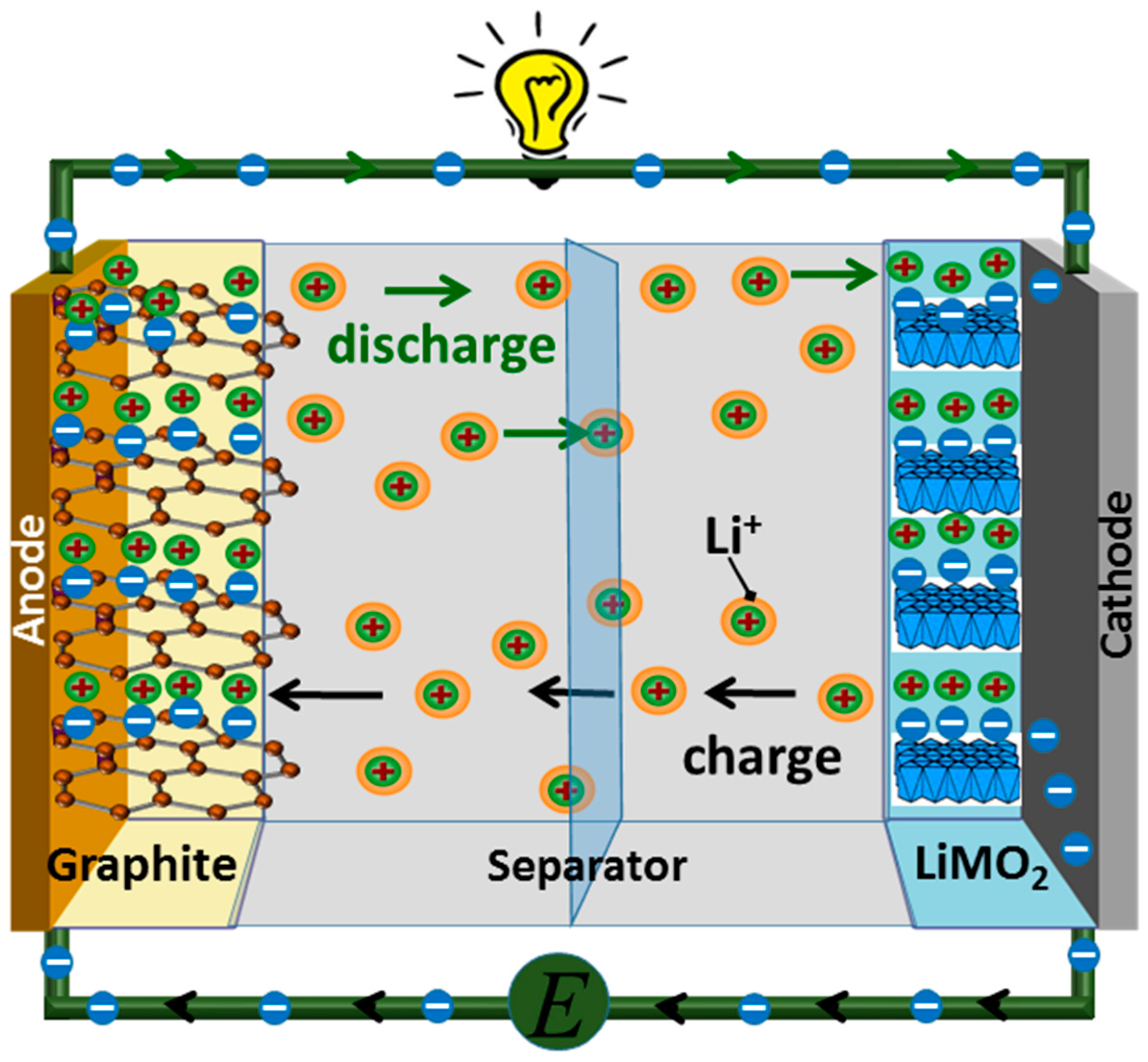
A new all-solid-state lithium battery can not only store nearly twice as much energy as a standard lithium-ion battery, but it’s also not prone to catching fire like its present-day, commercial counterparts.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
